Abstract
Background: Between 50-65% of myelofibrosis (MF) patients who undergo allogeneic hematopoietic cell transplantation (allo-HCT) are cured. Patient age, performance status, comorbidities, donor type, and HLA compatibility are established risk factors for allo-HCT outcomes. However, prognostic information after transplant in MF is largely based on JAK2-mutated patients, as this molecular abnormality is by far the most common and the first to be described. Since there is evidence suggesting that CALR-mutated patients could have a better outcome (Kroger et al., BBMT 2017), a study evaluating prognostic factors in this specific molecular subtype may be of clinical interest.
Method: This is a retrospective, registry-based study approved by the Chronic Malignancies Working Party (CMWP) of the European Society for Blood and Marrow Transplantation (EBMT). For the present study, inclusion criteria were adult patients with CALR-mutated MF undergoing first allo-HCT between 2005 and 2019. Patients transplanted from umbilical cord blood or with history of leukemic transformation were excluded. Primary study outcomes were OS, progression-free survival (PFS), relapse, non-relapse mortality (NRM) and graft-versus-host disease (GVHD)-free and relapse-free survival (GRFS).
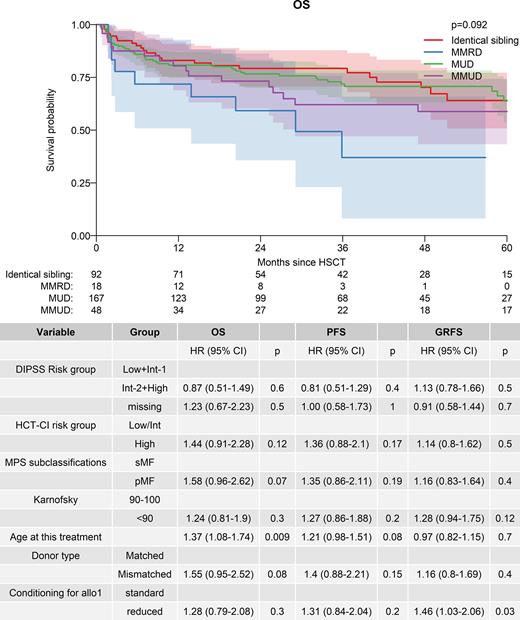
Results: 346 patients transplanted in 123 centers were included, of whom 207 (59.8%) were male. A total of 244 (70.5%) patients had primary MF (PMF) and 102 patients had MF secondary to prior essential thrombocythemia (SMF). Median interval from diagnosis to allo-HCT was 50.9 months (IQR: 17.2-155.6). Median age was 57.4 years (IQR: 51.2-63). DIPSS score at time of allo-HCT was available in 256 (74%) patients and was high, intermediate-2, intermediate-1 and low in 17%, 46%, 35%, and 2%, respectively. Sixty-nine patients (21%) had a high comorbidity index (HCT-CI) > 3. Seventeen patients (7.5%) had been splenectomized before transplant. Reduced intensity (RIC) regimens were utilized in 63% of patients and the donor source was peripheral blood in 95% of cases. Regarding donor type, an HLA-matched sibling donor (MSD), mismatched related donor (MMRD), matched unrelated donor (MUD) and mismatched unrelated donor (MMUD) was used in 92 (27%), 18 (5%), 167 (48%) and 48 (14%) of patients, respectively. T cell depletion was used in 77% of patients. Median time to neutrophil engraftment was 17 days. Incidence of acute GVHD grades II-IV at 100 days and extensive chronic GVHD at 60 months were 27% and 28%, respectively. After a median follow-up of 40 months from transplant, the estimated 5-year OS rate, PFS, GRFS, NRM and relapse incidence was 63%, 57%, 35%, 25% and 18%, respectively. Causes of death were relapse/progression (n=11), GVHD (n=36), infection (n=27), organ failure/toxicity (n=11), secondary malignancy (n=6), other (n=9), and unknown (n=3). The estimated 3-year OS rate was 79%, 37%, 72% and 62% for allo-HCT from MSD, MMRD, MUD and MMUD, respectively (p=0.09; Figure 1). The estimated 5-year OS rate in patients transplanted from an HLA identical donor was 64%. In univariable analysis, a high comorbidity index (47% vs. 66%; p=0.013) and a PMF diagnosis (55% vs. 79%; p=0.008) correlated with inferior 5-year OS rate. These two factors were associated with higher NRM rate at 5 years after transplant (HCT-CI > 3: 28% vs. 19%, p=0.033; PMF: 31% vs. 12%, p=0.012), with no difference in relapse incidence. Table 1 shows the results of the multivariable analyses for the main outcomes. As can be seen, older age (HR: 1.37, 95% CI: 1.08-1.74; p=0.009) was the only factor significantly associated with a worse OS, whereas PMF (HR: 1.58, 95% CI: 0.96-2.62; p=0.07) and transplant from a mismatched donor (HR: 1.55, 95% CI: 0.95-2.52; p=0.08) had a near-to-significant trend for decreased OS.
Conclusion: this is the largest series of allo-HCT for CALR-mutated MF patients. Overall, the results seem favorable, especially in patients with SMF or transplanted from an HLA-identical donor, where the 5-year OS in this multicenter series was 79% and 64%, respectively. NRM was the main cause of treatment failure.
Figure 1. Overall survival after transplant in 346 CALR-positive myelofibrosis patients depending on the donor type.
Table 1. Results of the multivariable analyses for the main study outcomes.
Disclosures
Kröger:Takeda: Consultancy, Honoraria; Sanofi: Honoraria; Kite: Honoraria; Neovii: Honoraria, Research Funding; Riemser: Research Funding; DKMS: Research Funding; Amgen: Honoraria; BMS: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Jazz: Honoraria. Robin:MEDAC, ASTEX: Research Funding. Finke:Riemser Pharma: Research Funding. Rambaldi:Celgene-BMS: Honoraria; Janssen: Honoraria; Roche: Honoraria; Incyte: Honoraria; Novartis: Honoraria; Kite-Gilead: Honoraria; Jazz: Honoraria; ABBVIE: Honoraria; Astellas: Honoraria; Pfizer: Honoraria; Amgen: Honoraria; Omeros: Honoraria. Drozd-Sokolowska:Sanofi: Honoraria; Janssen: Honoraria; Servier: Honoraria, Speakers Bureau; AbbVie: Honoraria, Speakers Bureau; Roche: Consultancy. Polverelli:Novartis Pharma: Consultancy. Yakoub-Agha:Bristol Myers Squibb: Honoraria; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria; Kite, a Gilead Company: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Support. McLornan:CELGENE BMS: Research Funding, Speakers Bureau; ABBVIE: Speakers Bureau; JAZZ: Honoraria, Speakers Bureau; NOVARTIS: Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.